Gastroparesis Marketed and Pipeline Drugs Assessment, Clinical Trials and Competitive Landscape
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
The gastroparesis pipeline consists of 17 pharmaceuticals spanning all stages of development, with approximately 90% of the drugs in early-stage development. Antagonists targeting the D2 receptor are expected to remain an important mechanism of action within the gastroparesis space. The report provides a data-driven overview of the current and future competitive landscape in gastroparesis therapeutics.
Gastroparesis is a chronic disease characterized by delayed gastric emptying of solid food in the absence of mechanical obstruction of the stomach. Patients with gastroparesis experience chronic symptoms with episodes of exacerbation, which typically include postprandial fullness, nausea, vomiting, and upper abdominal pain. Patients may also present with gastroparesis-like symptoms without delayed gastric emptying, complicating the identification of patients with the disease. The overlap of upper gastrointestinal symptoms makes the distinction between gastroparesis and other disorders, such as functional dyspepsia, more challenging.
Gastroparesis is more than twice as common in females as in males. Caucasians have the highest prevalence of any type of gastroparesis across all ages, followed by African Americans. Diabetic gastroparesis patients had a significantly higher risk of mortality than those with idiopathic gastroparesis in the UK. Gastroparesis is largely undiagnosed because many patients with diabetic gastroparesis are asymptomatic.
What is the marketed drugs overview of the gastroparesis therapeutics market?
Current drug therapies used for the treatment and management of gastroparesis include prokinetics and antiemetics.
Prokinetic agents are the cornerstone of pharmacotherapy for gastroparesis. Metoclopramide (Reglan and Gimoti), a dopamine D2 receptor antagonist, has both prokinetic and antiemetic properties. It is the only medication specifically approved by the FDA for the treatment of gastroparesis. It increases the contraction amplitude of esophageal fundic and antral musculature. Since it crosses the blood-brain barrier, the use of metoclopramide is often limited by its undesired side effects. These range from mild sedation and agitation to extrapyramidal effects like involuntary movements.
Antiemetics are often used for symptomatic control of nausea and are often considered as first-line therapy for gastroparesis. Antiemetic agents such as 5-hydroxytryptamine 3 (5-HT3) receptor antagonists (ondansetron and granisetron), phenothiazines (prochlorperazine and chlorpromazine), and antihistamines (promethazine, dimenhydrinate, and cyclizine) are frequently used for the symptomatic treatment of gastroparesis.
Other less frequently used therapies include nortriptyline, ghrelin agonists, and cannabinoids or cannabis-related medications. Several studies have evaluated botulinum toxin injection, but these studies were often unblinded and had small numbers of patients.
What is the pipeline drugs overview of the gastroparesis therapeutics market?
R&D activities for the treatment of gastroparesis are very limited, with a total of one molecule present in the current gastroparesis pipeline. There are two drugs in the late-stage (Phase III) gastroparesis pipeline.
Dopamine receptor has emerged as a key target for most pipeline candidates within the gastroparesis space. After the dopamine receptor, the other most targeted mechanisms of action (MOAs) under evaluation are 5-HT4 and G protein-coupled receptor 38.
Most molecules in the gastroparesis late-stage pipeline are being developed by mid-sized pharma/biopharma companies. The main route of administration of the late-stage molecules is oral. Molecules in development for gastroparesis have slightly higher PTSR at Phase I when compared to the overall gastrointestinal therapy area. At Phase II and Phase III, gastrointestinal molecules have a higher PTSR than gastroparesis pipeline molecules.
Other targets currently being investigated in the pipeline include tetrahydrobiopterin, substance P receptor, cannabinoid receptor 1, corticotropin-releasing factor receptor 1, and sodium-dependent noradrenaline transporter.
Which are the key sponsors in the gastroparesis therapeutics market?
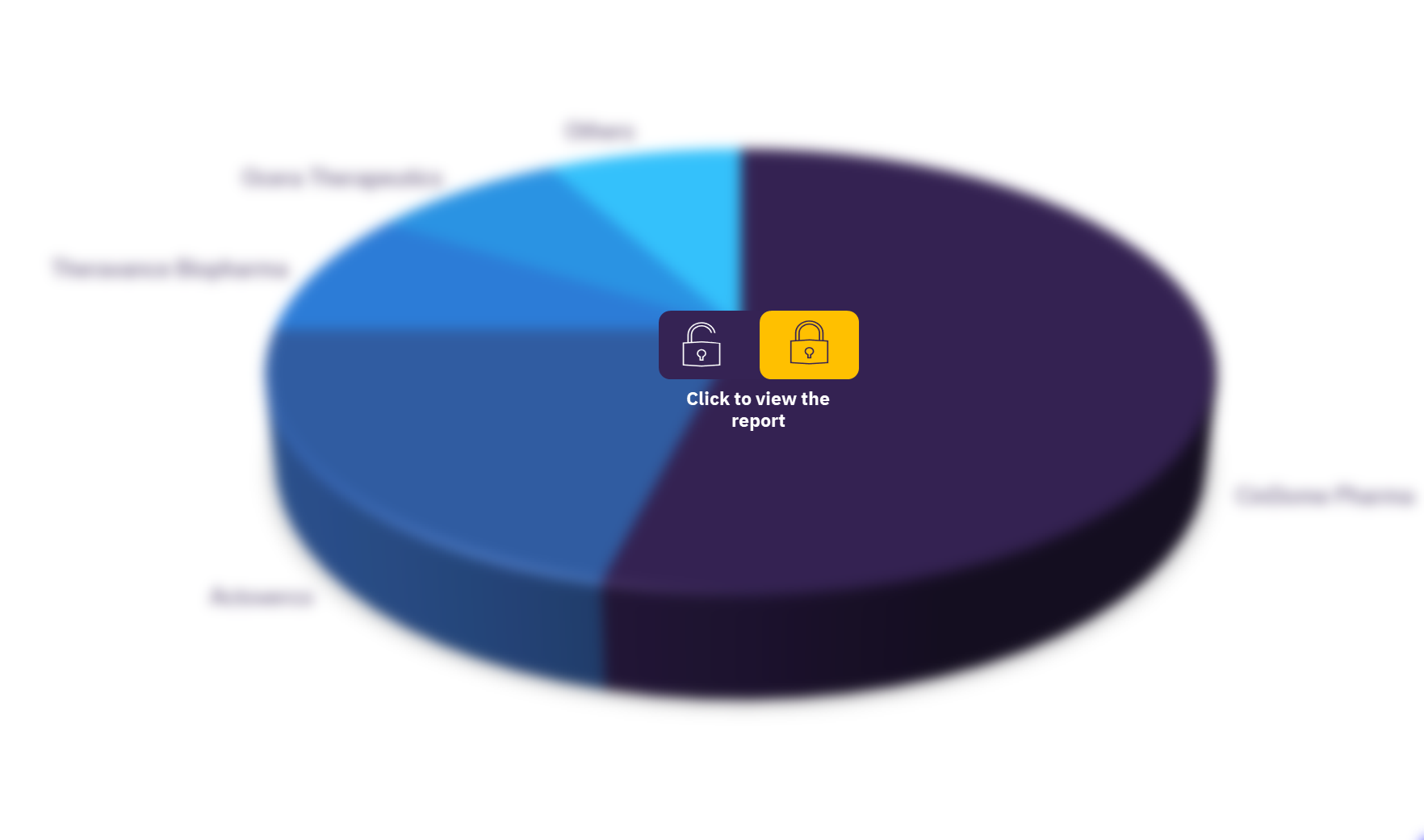
The key sponsors in the gastroparesis therapeutics market are Actoverco, Theravance Biopharma, Ocera Therapeutics, CinDome Pharma, Atoll Pharm, Vanda Pharmaceuticals, GlaxoSmithKline, Evoke Pharma, Allergan, and Takeda Pharmaceuticals. Takeda Pharmaceuticals is the top sponsor in gastroparesis clinical research followed by Allergan and Evoke Pharma.
Gastroparesis therapeutics market, by key sponsors
To know more about key sponsors, download a free report sample
Market Report Scope
| Key sponsors | Actoverco, Theravance Biopharma, Ocera Therapeutics, CinDome Pharma, Atoll Pharm, Vanda Pharmaceuticals, GlaxoSmithKline, Evoke Pharma, Allergan, and Takeda Pharmaceuticals |
Scope
GlobalData’s “Gastroparesis Marketed and Pipeline Drugs Assessment, Clinical Trials, Social Media and Competitive Landscape, 2021-2026” combines data from the Pharma Intelligence Center with in-house analyst expertise to provide a competitive assessment of the disease marketplace.
Components of the report include:
- Disease epidemiology
- Marketed drugs assessment
- Pipeline drugs assessment
- Clinical trials assessment
- Commercial assessment
- Social media overview
- Digital marketing overview
- The accompanying Excel file includes the following datasets:
- Marketed products exported from GlobalData’s Pharmaceutical Intelligence Center
- Pipeline-stage products exported from GlobalData’s Pharmaceutical Intelligence Center
- Upcoming catalysts in the gastroparesis market exported from the Catalyst Calendar on GlobalData’s Pharmaceutical Intelligence Center
- A list of patent expiry dates for gastroparesis products
Reasons to Buy
- Develop and design your in-licensing and out-licensing strategies through a review of pipeline products and technologies, and by identifying the companies with the most robust pipeline.
- Develop business strategies by understanding the trends shaping and driving the gastroparesis market.
- Drive revenues by understanding the key trends, innovative products and technologies, market segments, and companies likely to impact the global gastroparesis market in the future.
- Formulate effective sales and marketing strategies by understanding the competitive landscape and by analyzing the performance of various competitors.
- Identify emerging players with potentially strong product portfolios and create effective counterstrategies to gain a competitive advantage.
- Organize your sales and marketing efforts by identifying the market categories and segments that present maximum opportunities for consolidations, investments, and strategic partnerships.
Table of Contents
Frequently asked questions
-
Which are the key sponsors in the gastroparesis therapeutics market?
The key sponsors in the gastroparesis therapeutics market are Actoverco, Theravance Biopharma, Ocera Therapeutics, CinDome Pharma, Atoll Pharm, Vanda Pharmaceuticals, GlaxoSmithKline, Evoke Pharma, Allergan, and Takeda Pharmaceuticals.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.
Related reports
View more Pharmaceuticals reports